.svg)

Patient Experience Data on Health Care Gaps

Situation
Insmed, a biopharmaceutical company with a product registered for non-tuberculous mycobacterial lung disease (NTM-LD, a rare disease), sought to generate a deeper understanding of the burden of non-tuberculous mycobacterial lung disease (NTM-LD) and patients’ experiences with diagnosis, care, and daily life across Europe. Given the complexity of the disease, its overlap with underlying lung conditions, and the limited visibility of the patient experiences, there was a clear need for robust, patient-generated evidence to better understand unmet needs and identify opportunities to improve care.
Objective
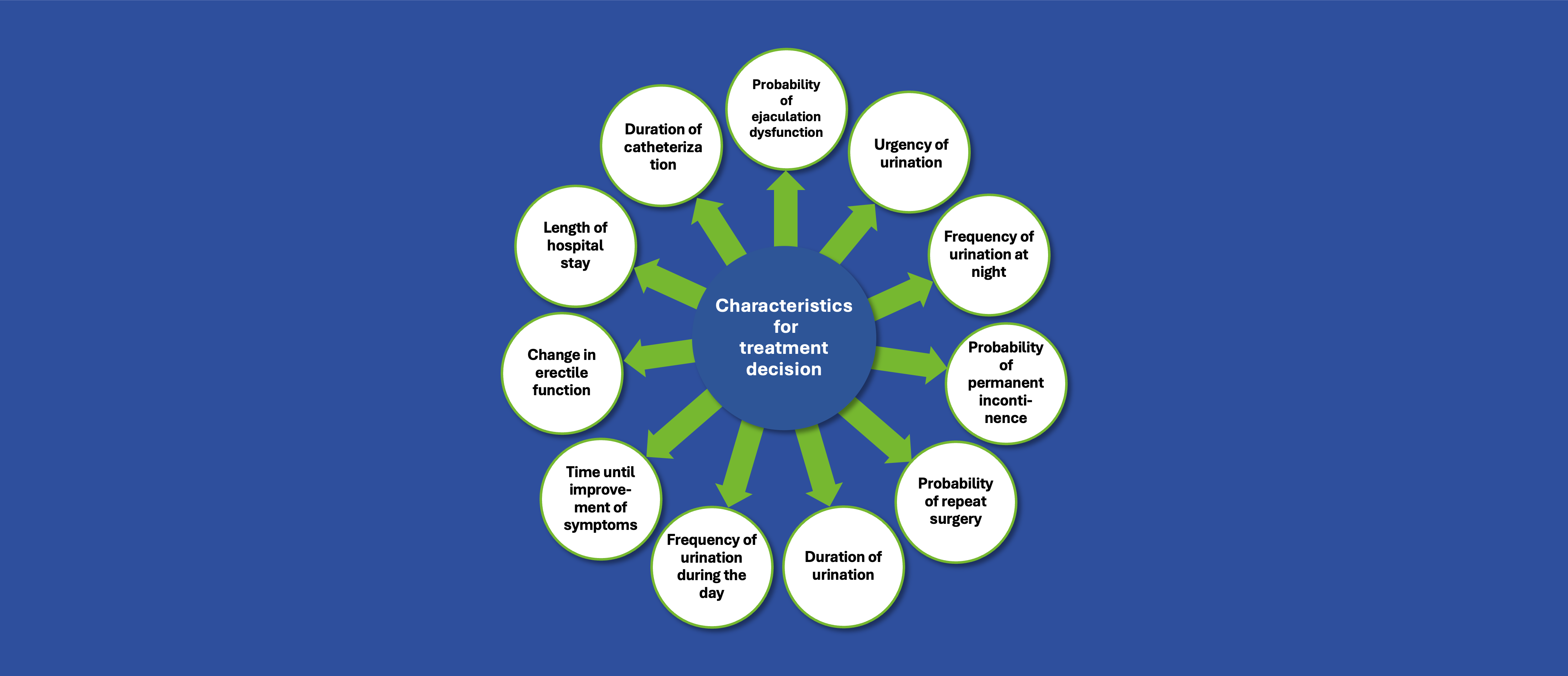
To design and implement a comprehensive European patient insights project involving more than 250 people living with NTM-LD. The project combined a quantitative online survey with semi-structured qualitative interviews to generate actionable evidence on disease burden and care experience with the aim of informing future patient-centred improvements in care.

Our Solution
Outcomes and Impact

“… I am extremely grateful for the exceptional service provided by admedicum … they went above and beyond to exceed my expectations … I highly recommend admedicum to anyone in need of their services.”
Conclusion
By executing the ENPADE project, admedicum generated high-quality patient evidence across eight European markets. The resulting insights gave Insmed a clear, patient-grounded understanding of disease burden, care gaps, and unmet needs, creating a strategic evidence base to guide future patient-centred activities and support improvements in NTM-LD care.
Related Case Studies