.svg)

Clinical Research & Development Through Patient Partnership

Situation
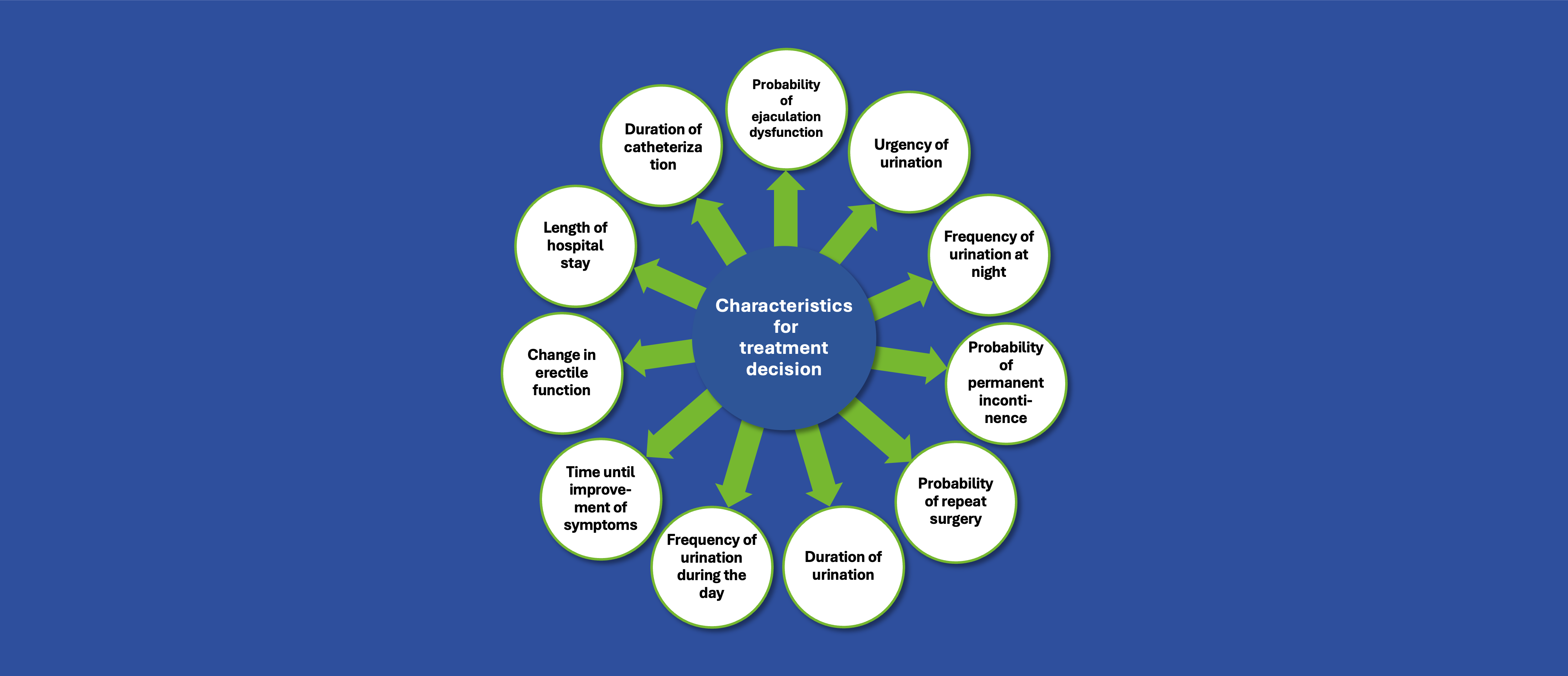
In the SUISSE MPC-2 study, a phase II clinical trial exploring a novel cell-based therapy for women with stress urinary incontinence, the teams from University of Zurich and MUVON Therapeutics, faced a unique set of challenges. The condition itself is often surrounded by stigma, making open discussion difficult for many women. In addition, the study’s design included strict eligibility criteria and was conducted at a single site, which further limited access for potential participants. Given the novelty of the investigated treatment, clear, honest, and empathetic communication was essential to build trust and encourage participation.
Objective
The objective was to break through barriers of awareness and trust and reach potential eligible patients who might benefit from this therapy. Recognizing that patients are not just study subjects but true partners in research, admedicum stepped in with its recruitment approach, a modular framework designed to support patients at every stage of their journey. This approach was built to ensure that individuals felt informed, understood, and empowered.

Our Solution
Outcomes and Impact

“What we do not research, remains hidden: Incontinence in women deserves visibility and scientific attention.”

"We are delighted to have engaged with admedicum early in the process of preparing our clinical trial ... . The admedicum team’s strategic and operational know-how as well as their legal and regulatory expertise provided a seamless service not only for patients but also for us as a Sponsor resulting in a streamlined and efficient recruitment process."
Conclusion
admedicum contributed to a seamless and efficient recruitment process by combining patient engagement expertise with strategic, operational, and regulatory know-how. Through these combined efforts, the clinical study turned recruitment into an efficient and collaborative process. By addressing the voices and concerns of women with stress urinary incontinence, patient-focused engagement broke down barriers, built trust, and encouraged participation.
Related Case Studies