.svg)

Patient Group Training on Marketing Authorization and HTA

Situation
A pharmaceutical company developing a new therapy for a rare neuromuscular disease recognized that patient organizations wanted to play a more active role in upcoming access discussions, but often lacked the knowledge and confidence to engage in marketing authorization and HTA processes. To support meaningful involvement, there was a need for a training approach that translated complex regulatory and market access topics into clear, relevant, and practical guidance for patient representatives.
Objective
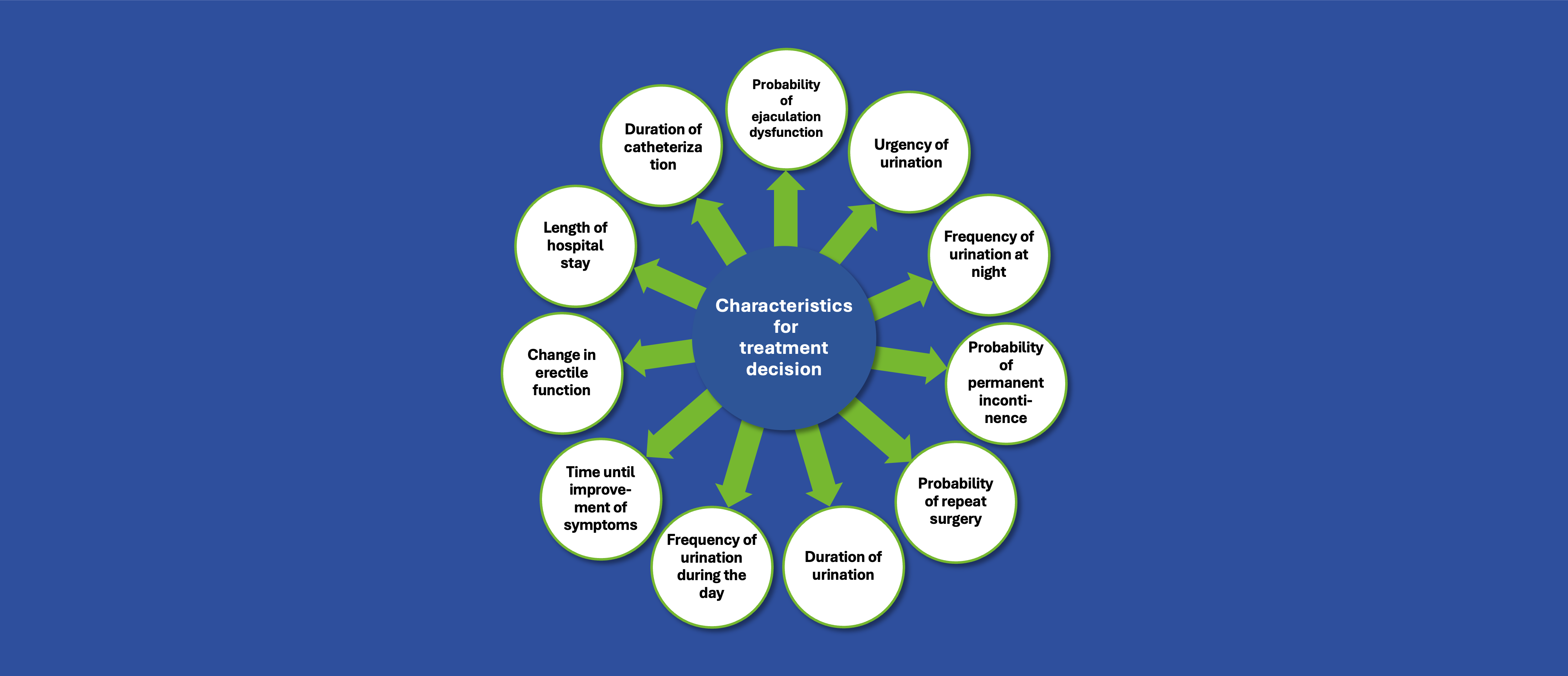
To design and deliver a comprehensive yet accessible HTA training program for European patient organization representatives, enabling them to better understand marketing authorization and market access processes and prepare for meaningful involvement in upcoming country-specific HTA discussions, in full compliance with applicable requirements.

Our Solution
Outcomes and Impact

“It was a great pleasure to meet you! Thanks again for the beautiful presentation. It was really useful! :) We will try to apply and explore the opportunities mentioned.”

“Thanks for the good and interactive training today. This helps us as an association in our empowerment and influence. Thank you also for sending the presentation. If there are any issues, I will be happy to contact you.”
Conclusion
admedicum translated complex regulatory and HTA concepts into a practical, patient-friendly training experience that empowered patient organizations to engage more confidently and effectively. By combining subject-matter expertise with strong patient engagement know-how, admedicum helped build the capabilities needed for more meaningful patient involvement in future marketing authorization and market access processes.
Related Case Studies